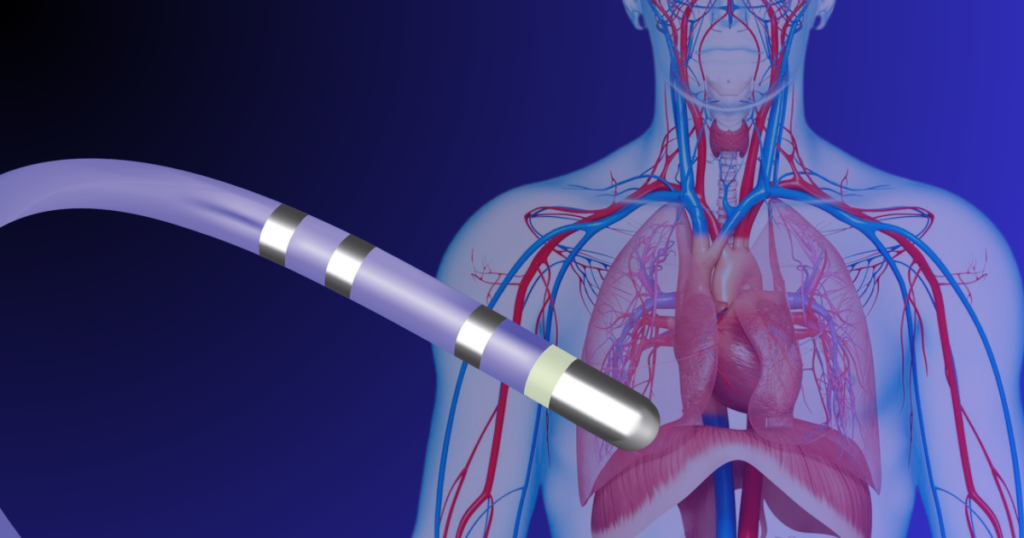
The NHS has begun trialing pulsed field ablation (PFA), a next-generation cardiac ablation therapy for treating atrial fibrillation. The move marks an important step in the UK adoption of advanced electrophysiology catheter technologies designed to improve procedural safety and efficiency.
According to recent BBC reporting, UK hospitals are now evaluating PFA as a non-thermal alternative to conventional ablation methods.
Manufacturing the Complex Catheters Behind PFA Therapy
PFA catheters represent one of the most sophisticated classes of minimally invasive cardiovascular devices. These electrophysiology catheters integrate fine conductors, electrodes, multi-lumen shafts, and advanced polymer materials within tightly controlled geometries.
Producing these assemblies requires precision, non-contact manufacturing processes capable of preparing delicate materials without mechanical stress or conductor damage.
Laser processing is widely used in advanced catheter manufacturing for applications including:
-
Fine wire stripping for electrode terminations
-
Selective material removal on catheter components
-
Preparation of micro-coax and multi-conductor cables
-
Precision processing of polymer-insulated medical wires
Laser Wire Solutions supplies laser processing systems and production-ready component supply used by medical device manufacturers producing PFA and other advanced cardiac catheters.
UK-Based Manufacturing Expertise Supporting Global MedTech
The introduction of PFA into NHS clinical evaluation highlights the UK’s role in adopting next-generation cardiac therapies. It also reflects the global manufacturing ecosystem required to bring complex catheter technologies from development into clinical use.
Laser Wire Solutions, headquartered in Wales, United Kingdom, supports major cardiovascular and electrophysiology device manufacturers worldwide through precision laser processing used in catheter component preparation.
By enabling repeatable, scalable manufacturing of critical catheter elements, laser-based processes help medical device companies transition therapies such as pulsed field ablation from R&D into validated production and clinical deployment.
Scaling Production for the Next Wave of Cardiac Ablation Technologies
As pulsed field ablation adoption expands globally, manufacturers face increasing demand for high-yield production of complex electrophysiology catheters. Automated, validated laser processes play a key role in achieving the consistency required for regulatory approval and large-scale manufacturing.
Laser Wire Solutions continues to work closely with medical device engineering and manufacturing teams to support the development, scale-up, and production of advanced catheter technologies – including those used in pulsed field ablation therapies now being evaluated within the NHS.